
October 23, 2020
PHC Corporation
2-38-5 Nishishimbashi, Minato-ku, Tokyo 105-8433, Japan
JCR Pharmaceuticals Co., Ltd.
3-19 Kasuga-cho, Ashiya, Hyogo 659-0021, Japan
Tokyo and Hyogo, October 23, 2020 - PHC Corporation (headquarters: Minato-ku, Tokyo, President: Kyoji Morimoto, hereafter PHC) and JCR Pharmaceuticals Co., Ltd. (headquarters: Ashiya, Hyogo, Chairman and President: Shin Ashida, hereafter JCR) announce the release of Melon Nikki™*1, a dedicated smartphone app for GROWJECTOR® L*2, a motorized growth hormone delivery device that is designed to help improve medication adherence among patients using injectable growth hormone therapy in Japan.
Melon Nikki™ is a medication management app for growth hormone therapy that was jointly developed by PHC and JCR. It has been provided for use in clinical research (Project Leader: Dr. Tatsuhiko Urakami of Nihon University Hospital), starting February 2019, that was designed to investigate the factors associated with low medication adherence of growth hormone therapy and to gather information about the practical use of the Melon Nikki™. The app has been determined ready for practical use, following the completion of the analysis of verification-test data, and it is now available on Google Play*3.
Growth hormone therapy for short-stature children requires continual subcutaneous injections and maintaining medication adherence is essential. Young patients are often scared of injections and administering injections can cause families considerable burden, raising concerns about medication adherence and negative impact on patient quality of life. There is a need for solutions that can reduce patient stress related to injections in order to improve adherence, while also being able to provide more convenient medical services for healthcare professionals and families.
GROWJECTOR® L is a motorized drug delivery device manufactured and sold by PHC, to be used exclusively for the recombinant human growth hormone GROWJECT® subcutaneous 6mg and 12mg*4 treatment which is manufactured and sold by JCR. The device can record patient injections (dosing history), which are stored in the device memory. Using the Melon Nikki™ app, the dosing history stored in GROWJECTOR® L can be seamlessly sent via Wi-Fi to a smartphone, allowing patients to easily check detailed dosing history including injection time and frequency without the need for manual input. Patient height and weight data saved in the app enables users to generate a growth curve that can be stored in the cloud along with their dosing history. The patient and their family can check on treatment status and their growth record at any time, as well as share information with doctors during consultations through their smartphone device screen. The app also has a fun feature*5 designed to reduce injection anxiety and therefore improve medication adherence.
Hiroyuki Tokunaga, Member of the Board and Director of the In Vitro Diagnostics Business Unit of PHC says, “Since we began developing blood glucose monitoring systems in 1991, we have developed, manufactured, and sold easy-to-use and highly accurate testing/analysis equipment. The third generation GROWJECTOR® L launched in 2017 has also been well-received by our customers for its precise technology and superior quality. With the launch of this dedicated medication management app, Melon Nikki™, we expect to reduce the stress of subcutaneous injections for patients and their families and enable more effective medication treatment by providing a solution that helps track dosing history. As we integrate our expertise in precision engineering with digital technology, we will contribute to better healthcare by providing best-in-class healthcare solutions that improve the quality of life for patients and respond to the needs of healthcare professionals.”
Toru Ashida, Vice President and in charge of Corporate Strategy of JCR says, “Since launching the motorized growth hormone delivery device GROWJECTOR® in 2006, we have carried out activities to provide product information as well as conducted research and development to meet the needs of patients undergoing treatment with recombinant human growth hormone GROWJECT®. The newly released Melon Nikki™ designed specifically for users of GROWJECTOR® L will not only support self-injections in growth hormone therapy, but also has the potential to improve patient’s medication adherence and information sharing with healthcare professionals. We expect it to become a good example of healthcare digital transformation in this age of IoT by making growth hormone therapy more effective. We will continue to provide a wide range of options in growth disorder treatment and contribute to the development of pediatric care.”
 Melon Nikki™ Website: jcrgh.com/melonnikki/top.html (Japanese website)
Melon Nikki™ Website: jcrgh.com/melonnikki/top.html (Japanese website)
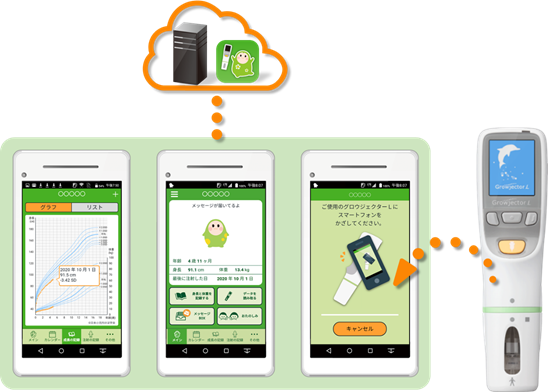 Using Melon Nikki™
Using Melon Nikki™
* Melon Nikki, GROWJECT, and GROWJECTOR L are registered trademarks or trademarks of JCR Pharmaceuticals Co., Ltd.
*1 Melon Nikki™ is available for Android (NFC Type B)OS. Compatible with all GROWJECTOR® devices with no updates required. Expected to be available for iOS devices in the future. This is not a medical app.
*2 Official generic term: Drug/vaccine injector, authentication number 228AABZX00109000
*3 Google’s digital downloading service offering applications, movies, music, books, etc. The Melon Nikki™ app can be downloaded for free.
*4 Approved indications of GROWJECT® 6mg/GROWJECT®12 mg:
- Growth hormone deficiency
- Turner syndrome
- Adult growth hormone deficiency
- Small for gestational age
Visit JCR website for further information: jcrgh.com/index.html (Japanese website)
*5 Young patients can choose clothes and other items for their avatars, as well as communicate with other young patients via the avatar about their daily treatments.
About In Vitro Diagnostics (IVD) Business Unit, PHC Corporation
Established in 1969, PHC Corporation is a Japanese subsidiary of the PHC Group, a global healthcare company that develops, manufactures, sells, and services solutions across diabetes management, life sciences and diagnostics, as well as providing healthcare services. Committed to its corporate mission, “We contribute to the health of society through our diligent efforts to create healthcare solutions that have a positive impact and improve the lives of people,” the company offers value-added products and services to customers in over 125 countries. The IVD Business Unit has worked to improve medical services for patients by contributing to early diagnosis and effective medical care for diseases such as diabetes and asthma through development, manufacturing, and distribution of testing and analyzing devices including blood glucose monitoring systems.
www.phchd.com/global/phc/profile
About JCR Pharmaceuticals Co., Ltd
JCR is a specialty pharma company engaged in research, development, manufacturing, and marketing of biopharmaceuticals and regenerative medicine with a focus on rare diseases. Its philosophy, “Contributing towards people’s healthcare through pharmaceutical products” drives JCR to create innovative pharmaceutical products as value-added treatment options for the under-served patient populations.
www.jcrpharm.co.jp/en/site/en/index.html
Media contact
Corporate Communications Group
PHC Holdings Corporation
+81-3-6778-5311
Investors and Media
JCR Pharmaceuticals Co., Ltd.
Email: ir-inrfo@jcrpharma.co.jp